Coronavirus (COVID-19) remains a powerful force in your daily lives—and in ours
At Hillrom, we are ready to deliver the critical products, services and solutions you need to care for your patients in these challenging times. In fact, we’ve more than doubled production of our most in-demand products to help hospital and primary care clinicians like you treat COVID-19 patients in the following areas:
FRONT LINE SCREENING & DIAGNOSTIC CAPACITY
We’re maintaining availability of thermometry, vital signs and resting ECG devices to help you quickly examine patients and determine the level of care they need.
INFECTION CONTROL & REDUCTION OF CROSS-CONTAMINATION
We’ve increased production of disposable thermometry probe covers, single-use blood pressure cuffs and other solutions to help you triage patients, dedicate supplies to isolation rooms and minimize the risk of cross-contamination.
RESPIRATORY DISTRESS MONITORING, NON-INVASIVE VENTILATION & AIRWAY CLEARANCE THERAPY
We’re delivering innovative technologies to help you continuously monitor respiration rate. And for your patients in respiratory distress, we’re offering solutions to support their breathing and help move secretions out of their lungs.
INFRASTRUCTURE EXPANSION TO CREATE SURGE CAPACITY
We’re working with governments and hospital systems to prepare to treat, monitor and move more patients in acute settings—and automatically detect signs of possible deterioration.
CARE TEAM COMMUNICATIONS EXTENSION
We’re helping care teams stay connected across temporary spaces—from hospitals and adjacent temporary facilities to homes or remote working spaces.
As global demand has grown substantially for several of our critical products, our business operations are continuing with no material interruption. We are focused on supporting these pressing needs—and, as always, supporting caregivers like you.
New COVID-19 Innovations
Supporting Caregivers in the Fight Against COVID-19
Supporting Caregivers in the Fight Against COVID-19

Outfitting a Field Hospital in Kuwait—Fast
In just five days, Hillrom worked with Kuwait's Advanced Technology Company and the Kuwait MOH to outfit a field hospital with Hillrom beds, stretchers, vital signs monitors and respiratory care products for its 40 ICUs, 200 ward rooms and 19 ED rooms.
Supporting Caregivers in the Fight Against COVID-19
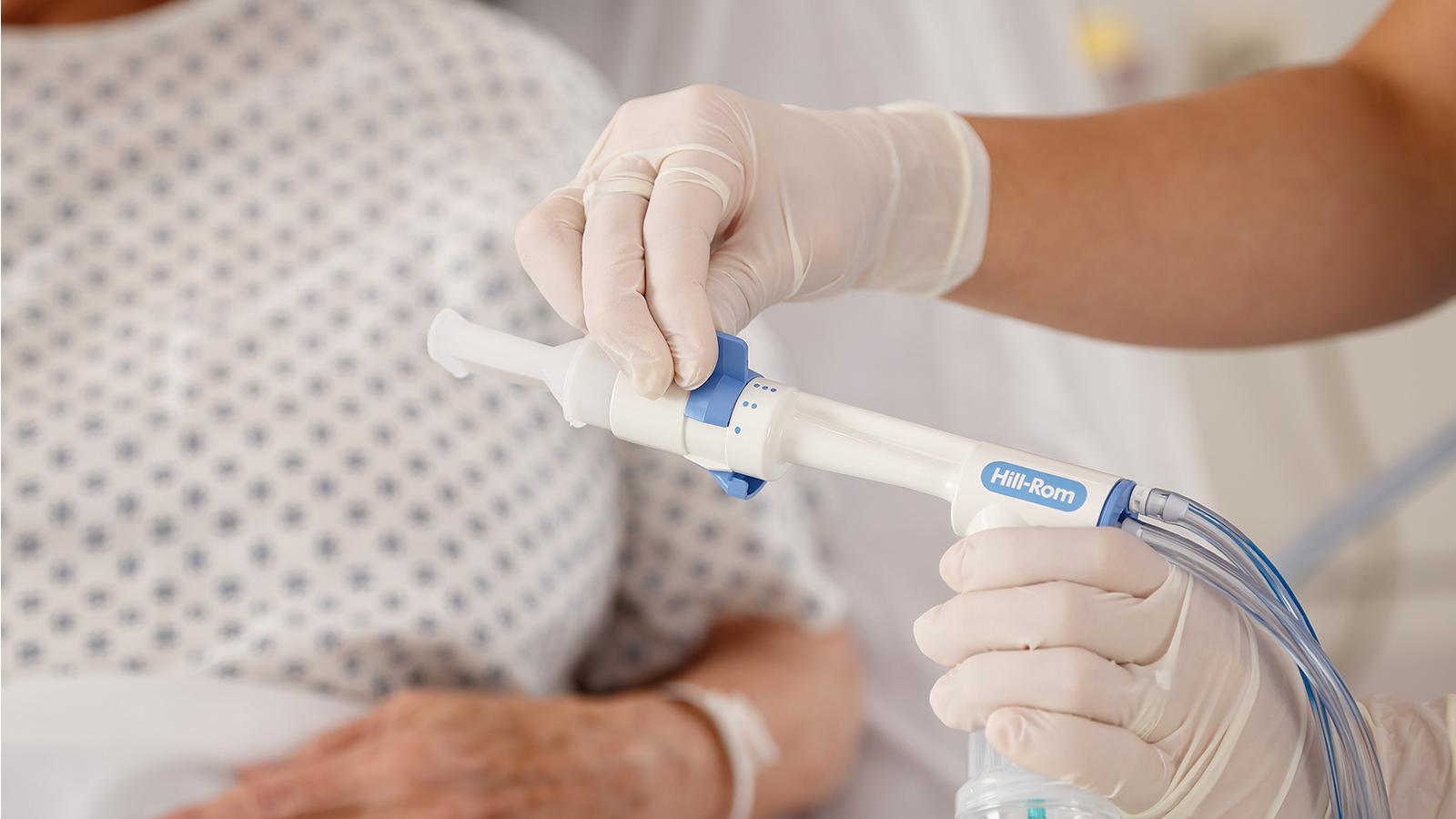
The MetaNeb System Was Granted FDA Emergency Use Authorization (EUA) for COVID-19
Data suggests that up to 30% of COVID-19 patients with severe disease have significant pulmonary mucus production. In this news brief, the team at Emory University in Atlanta discusses the use of Oscillation and Lung Expansion (OLE) with COVID-19 patients.
Supporting Caregivers in the Fight Against COVID-19

Flex Up Your ICU Capacity
What if you could “flex up” your med-surg units to increase capacity, not only for COVID-19 care but also for future situations where patient demand is unpredictable? It is possible, and flexible solutions from Hillrom can help.

